December 06, 2018
Infant ibuprofen has been recalled from shelves at CVS, Family Dollar and Walmart over concerns about dosing.
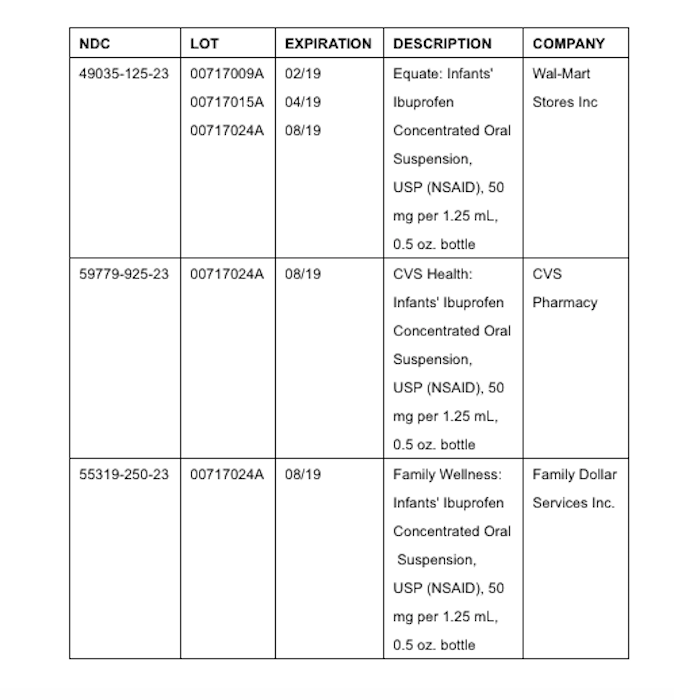
Three recalled lots of "Concentrated Oral Suspension, USP (NSAID) 50 mg per 1.25 mL" may have concentrations of ibuprofen that are too high and therefore could be dangerous to infants, New Jersey drug manufacturer Tris Pharma announced on Thursday.
The over-the-counter liquid pain reliever and fever reducer is meant for infants between the ages of six and 23 months old. Adverse events from the medication may include nausea, vomiting, upper abdominal pain, diarrhea, gastrointestinal pain, ringing in the ears and headache. No adverse events related to the recalled ibuprofen have been reported to date, however, according to Tris Pharma.
Additionally, the announcement states: "There is a remote possibility that infants, who may be more susceptible to a higher potency level of drug, and therefore may be more vulnerable to permanent NSAID-associated renal injury.”
RELATED READ: Woman gives birth via uterus transplanted from deceased donor — the world's first
The detailed information for the affected recalled product, which were packaged in half-ounce bottles, is below:
Tris Pharma sold the affected lots of ibuprofen to one customer, which distributed the product to some U.S. retailers. Tris Pharma notified its customer by urgent recall notice and is arranging for the return of the recalled product, it said.
Consumers who have the medication should return it or throw it away, and retailers should discontinue selling it.